A group led by Ursula Gompels from the London School of Hygiene & Tropical Medicine, University of London, did next generation sequencing on three ciHHV6A cardiac patients and found superinfections of HHV-6A in two of the three. They characterized the first full genome sequence of ciHHV-6A and demonstrated that the inherited ciHHV6 genome was similar to, but distinct from known exogenous (community acquired) strains of HHV-6A (Tweedy 2016).
Characterization of the integrated genome showed that it had all the features necessary to reactivate as an emerging virus (Tweedy 2016). This raises possibilities for studying new biomarkers related to gene expression or reactivation of the integrated virus in cases of disease.
In papers published last year, Gompels and Uwe Kuhl’s German cardiology group at Charite University, together with Dirk Lassner’s cardiac diagnostic group at IKDT, showed that the inherited virus genome demonstrated reactivity or gene expression and that anti-viral therapy could reduce the expression (Tweedy 2015, Kuhl 2015).
Bhupesh Prusty from the University of Würzburg has shown that microbial superinfection can reactivate ciHHV-6A replication in vitro through effects on telomere stability (Prusty 2013), and Nicola Royle from the University of Leicester found a link between disrupted telomere stability and ciHHV-6B genome release occurring sporadically in vitro (Royle 2014).
Peter Medveczky’s lab from the University of Florida has also published evidence of superinfection in four ciHHV6A/B in patients with cognitive dysfunction. In that case, gene expression was from the community acquired and not the inherited virus genome. They also showed that long term but not short-term antiviral therapy could inhibit HHV-6A replication (Pantry 2013).
Mary Caserta from the University of Rochester found that ciHHV-6A/B was negatively correlated with mental development in a group of infants. The effects of virus superinfection or reactivation are not yet known (Caserta 2014).
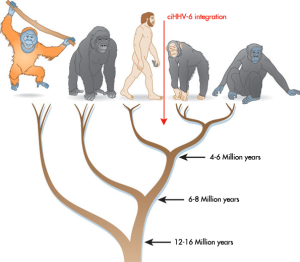
Both ciHHV6A and exogenous HHV-A have been associated with inflammatory and cardiovascular disease (Kuhl 2015, Tweedy 2015, 2016). The Gompels team determined prevalent integration sites at the telomere of human chromosome 17 and concluded that the integration of HHV-6A and HHV-6B took place after the divergence of humans from chimpanzees (Tweedy 2016). This has been estimated at between 2-6 million years ago.
An emerging vertically acquired infection from the past?
Read the full paper.