Little is known about the effects of the proteins HHV-6A/B produce during latency. Researchers, led by Kazuhiro Kondo, MD, PhD at Jikei University School of Medicine, identified a protein named SITH-1 that they believe greatly increases the risk of depression by prolonging hypothalamic-pituitary-adrenal (HPA) axis activation. The HPA axis hyperactivation for prolonged periods can lead to depressive symptoms.
In mice, expression of the HHV-6B SITH-1 protein was found to increase apoptosis in the olfactory bulb, thereby suppressing hippocampal neurogeneration, which activates the HPA axis and leads to depressive symptoms. In human astrocyte cell lines, SITH-1 was found to form a complex with CAML, a calcium modulator. The SITH-CAML complex increased the intracellular concentration of calcium enough to induce apoptosis in human astrocytes. To assess the prevalence of SITH-CAML in depressive patients, levels of anti-SITH-CAML antibodies were measured and compared to healthy controls. The positive rates of the antibodies were 79.8% in depressive patients and 24.4% in controls, with an odds ratio of 12.2. Anti-SITH-CAML antibodies were 77.7% accurate as a diagnostic marker for depression.
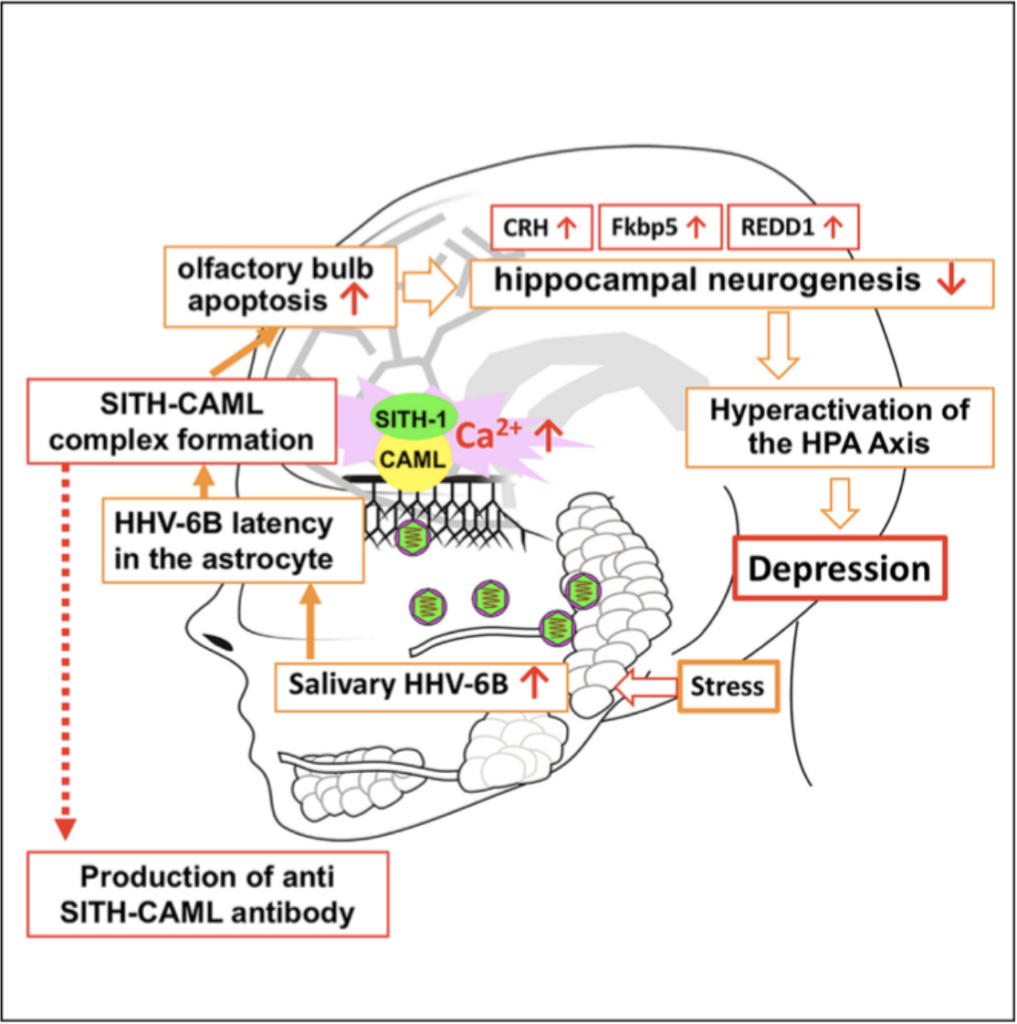 The research builds off Dr. Kondo’s previous research that found 88% of stressed office workers were positive for reactivated HHV-6 before the holidays, but only 23% were positive after the holidays. Looking for a potential mechanism, Kondo assessed SITH-1 dependent mental status changes and found that manic behavior could be induced by SITH-1 expression in glial cells in mice. He then reported that over half of depressed patients and three-quarters of bipolar patients were positive for anti-SITH-1 antibodies.
The research builds off Dr. Kondo’s previous research that found 88% of stressed office workers were positive for reactivated HHV-6 before the holidays, but only 23% were positive after the holidays. Looking for a potential mechanism, Kondo assessed SITH-1 dependent mental status changes and found that manic behavior could be induced by SITH-1 expression in glial cells in mice. He then reported that over half of depressed patients and three-quarters of bipolar patients were positive for anti-SITH-1 antibodies.
By demonstrating that HHV-6B can lately infect olfactory neurons, produce a unique protein that triggers apoptosis of hippocampal neurons, and induce prolonged activation of the HPA axis, Kondo et al have provided a novel groundwork for future research in depression. Additionally, they have identified a novel cellular pathway that may help researchers better understand the effects of latent herpesvirus infections that are highly prevalent throughout the population.
Read the full paper: Kobayashi 2020

