Viral dUTPases are strong inducers of activin A and enhance T follicular helper-cell differentiation.
Past studies from the laboratory of Maria Ariza and Marshall Williams at Ohio State University have found increased levels of autoanatibodies directed at human deoxyridine triphosphate nucleotideohydrolase (dUTPase) proteins (Halpin 2017). These proteins function as pathogen-associated molecular pattern proteins (PAMPs). When they engage toll-like receptor-2 (TLR2), they activate NF-κB which, in turn, upregulates many genes involved in innate and adaptive immunity.
A new study finds that in people with myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) there is a significant correlation between the presence of autoantibodies against the dUTPase proteins of HHV-6A and EBV and increased serum levels of activin A and IL-21. (Activin A belongs to the TGF-B superfamily.) These molecules are known to be important for T follicular helper (TFH) cell differentiation, and subsequent generation of high-affinity antibodies and of the long-lived plasma cells that produce them.
In vitro studies found the dUTPases were able to induce activin A, B cell-activating factor (BAFF) and other molecules important in TFH differentiation. They also induced CD4+ differentiation into IL-21 producing cells.
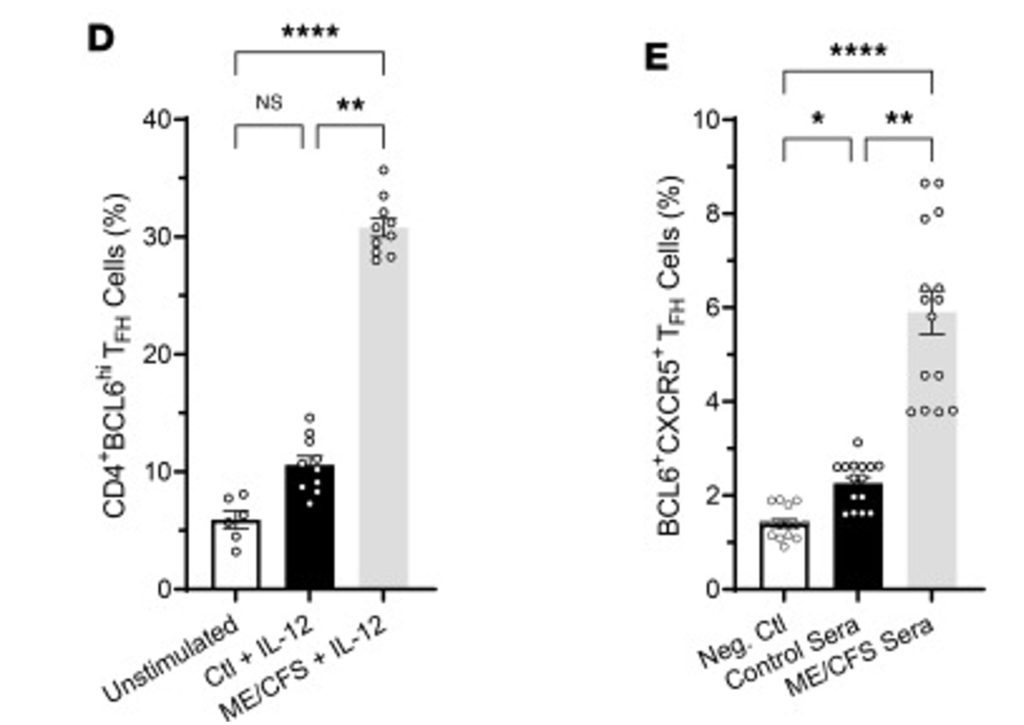
Figure 1. Effect of serum from people with ME/CFS and healthy control subjects in stimulating the differentiation of TFH cells.
In vivo studies in mice found that the EBV dUTPase induced the formation of splenic marginal zone B and invariant NKTFH cells. As shown in Figure 1, serum from people with ME/CFS, but not from control subjects, plus IL-12, induced the formation of CD4+BCL6hi TFH cells. And serum from people with ME/CFS, but not from control subjects, induced the formation of BCL6+CXCR5+ TFH cells.
These studies are consistent with previous studies, largely from this group, finding higher levels of autoantibodies to the dUTPases of both HHV-6A and EBV in people with ME/CFS. These studies also suggest that the dUTPases may encourage autoimmunity, and may be the circulating factors in serum that encourage the differentiation of TFH cells.
Read full article: Cox 2022

