Investigators in Burkina Faso conducted a study of 238 HIV-1 positive individuals to determine the prevalence of EBV and HHV-6 infections in the population, as well as the interactions between these viruses and the course of the HIV infection. The prevalence of HHV-6A and HHV-6B were similar, with no significant differences. As the epidemiology of HHV-6A remains unclear to a great extent, and it is infrequently found in the blood of healthy donors (Hall 2006, Alvarez-Lafuente 2004) in Europe and the USA, the results of this study shed light upon the variable geographical distribution of the two viruses and the potential effects of the higher rate of HHV-6A positivity on the health of those infected.
Using whole blood, the investigators tested for EBV-1, EBV-2, HHV-6A, and HHV-6B via qPCR and found that 13% of the population was positive for at least one of the viruses. 17 (7.1%) patients tested positive for HHV-6A and/or HHV-6B, and 10 (4.2%) had HHV-6A/B coinfections. Five patients had only HHV-6A, and two had only HHV-6B.
The prevalence of the two viral species in African cohorts has varied from studies of asymptomatic or symptomatic patient groups (Table 1). HHV-6B has been more commonly identified in symptomatic non-African cohorts (Table 2). HHV-6A was found to be the predominant variant in the serum of asymptomatic infants in a 2009 study (Bates 2009), while in symptomatic patients, similar to the Burkino Faso study, both HHV-6A and HHV-6B were found in Zambia at similar frequencies in both HIV negative (Kasolo 1997) and positive1 febrile children. The variation in the results of several studies points to differences in symptomatic or asymptomatic populations, detection methods, and sample types. For instance, Bates et al. found higher rates of HHV-6A in asymptomatic infants but more HHV-6B in febrile infants, which is consistent with the presumably higher incidence of primary HHV-6 infection among infants with fever. Moreover, the viral loads of the two species may confound the analyses depending on whether specific primers were used. Standardization of cohorts selected, detection thresholds, and other aspects of testing will allow for more accurate analysis.
Although the differences were not significant, HHV-6B was more often found in patients with a high HIV load, while HHV-6A was more common among those with low HIV loads. While EBV-2 was found significantly more often in patients with high CD4 T cell counts, the difference was not significant for HHV-6A and HHV-6B. Both species were more prevalent among patients who had received highly active antiretroviral therapy (HAART) compared to those who had not yet been treated. This too failed to reach statistical significance, but the results suggested that the therapy did not reduce herpesvirus infections. HAART treatment improves CD4 T cell restoration and lowers HIV viral load, and as CD4 T cells are targeted by HHV-6, this may allow for unimpeded HHV-6 replication. The exact role of HHV-6A or B in the disease progression of HIV+ individuals is unknown, although Emery and colleagues found an association of higher loads of HIV with HHV-6 (untyped) in tissues in a UK study (Emery 1999). HHV-6A/B is also able to induce the expression of CD4 on CD8 lymphocytes in vitro, thereby increasing the range of cells susceptible to HIV infection (Lusso 1991), but HHV-6A chemokine potently inhibits HIV infection (Catusse 2007) so the relationship with HIV/AIDS can be complex.
———–
1Kasolo, F.C. and Gompels, U.A. (2006) HHV-6 Genome: Similar and Different. In ‘Human herpesvirus-6’ 2nd Ed Elsevier:Oxford eds G. Krueger and D. Ablashi, pp 23-46.

Abbreviations- ND: not done, NA: information not available. Green shading represents the HHV-6 species found to be more prevalent in the cohort listed. Source and ©: HHV-6 Foundation 2017
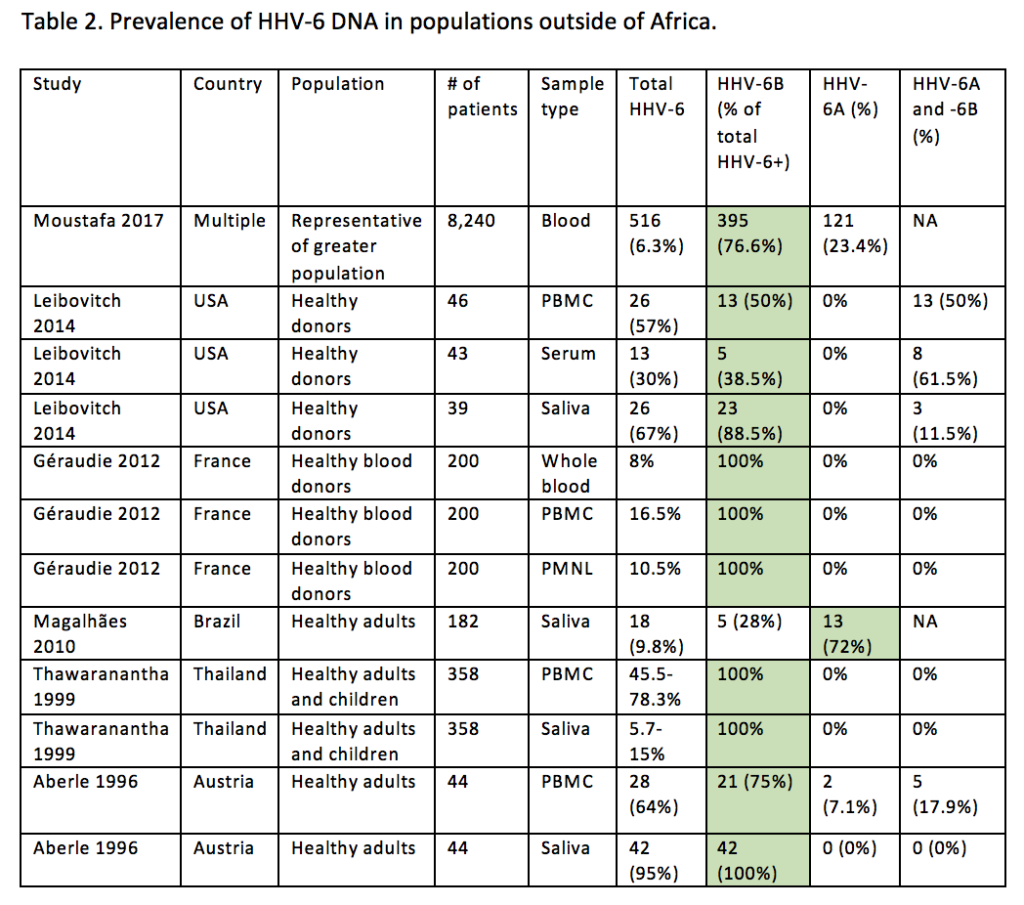
Abbreviations- NA: information not available. Green shading represents the HHV-6 species found to be more prevalent in the cohort listed. Source and ©: HHV-6 Foundation 2017