Oncogene activation, pro-inflammatory cytokines, genomic instability, expression of oncogenic miRNAs are induced by infection.
HHV-6A has been shown to infect thyroid cells (thyrocytes), and also has been linked to autoimmunity (Broccolo 2013) as well as Hashimoto’s thyroiditis (Caselli 2012, Sultanova 2017). People with Hashimoto’s thyroiditis are more likely than others to then develop thyroid cancer, although it is an unusual event. Also, Hashimoto’s thyroiditis shares with papillary thyroid cancer some molecular profiles such as, for example, the re-arrangement of the RET protooncogene (RET/PTC oncogene), the BRAF mutation, and activation of the phosphatidylinositol 3-kinase (PI3k) pathway.
There are three common types of thyroid cancer, each originating in follicular cells: well-differentiated (and least aggressive) papillary thyroid carcinoma (PTC), the more aggressive follicular thyroid carcinoma (FTC) and the very aggressive anaplastic thyroid carcinoma (ATC). There is evidence that thyroid cancers can start as the least aggressive form, and then develop into the more aggressive forms—driven by various pro-oncogenic forces.
Investigators from Sapienza University in Rome, Italy, investigated the possible role of HHV-6A in thyroid cancer. First, they infected a well-characterized papillary carcinoma thyroid cancer cell line, BCPAP, with HHV-6A and found it to be highly permissive. Infection of the cell line did not induce apoptosis nor produce a cytopathic effect. Rather, it produced a series of changes that all previously have been shown to be oncogenic:
- Increased secretion of pro-inflammatory cytokines, particularly IL-6, which may also sustain thyroid tumor cell growth and promote cancer progression, as shown in Figure 1;
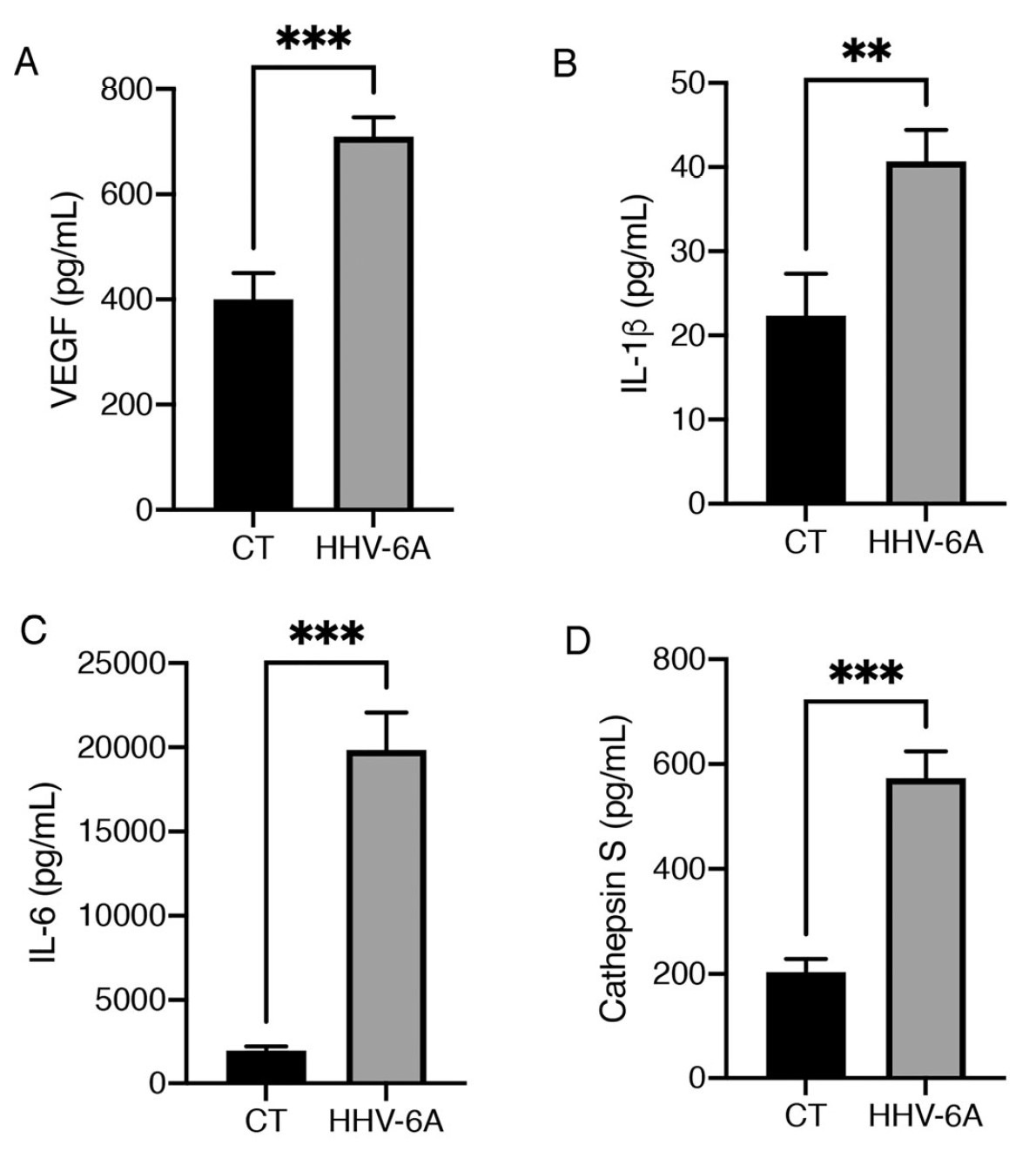
Figure 1. Levels of four cytokines following mock infection of cells (CT) or following infection with HHV-6A. ** indicates p < 01, *** indicates p < 0.001.
- Increased the production of VEGF, which enhances angiogenesis and tumor invasiveness, and also may suppress the immune response to malignant cells;
- Created genomic instability as reflected by the upregulation of the phosphorylated form of H2AX (H2AX);
- Increased the expression of PTEN, which has been reported to act as an oncogene in mutp53-carrying cells such as the BCPAP cell line;
- Induced an epithelial-to-mesenchymal transition (EMT), an important step in metastasis, as reflected in the upregulation of various mesenchymal markers, such as vimentin;
- Dysregulated the expression of several microRNAs, such as miR-155, miR-9, and the miR-221/222 cluster, which are involved in different steps of carcinogenesis, as shown in Figure 2.
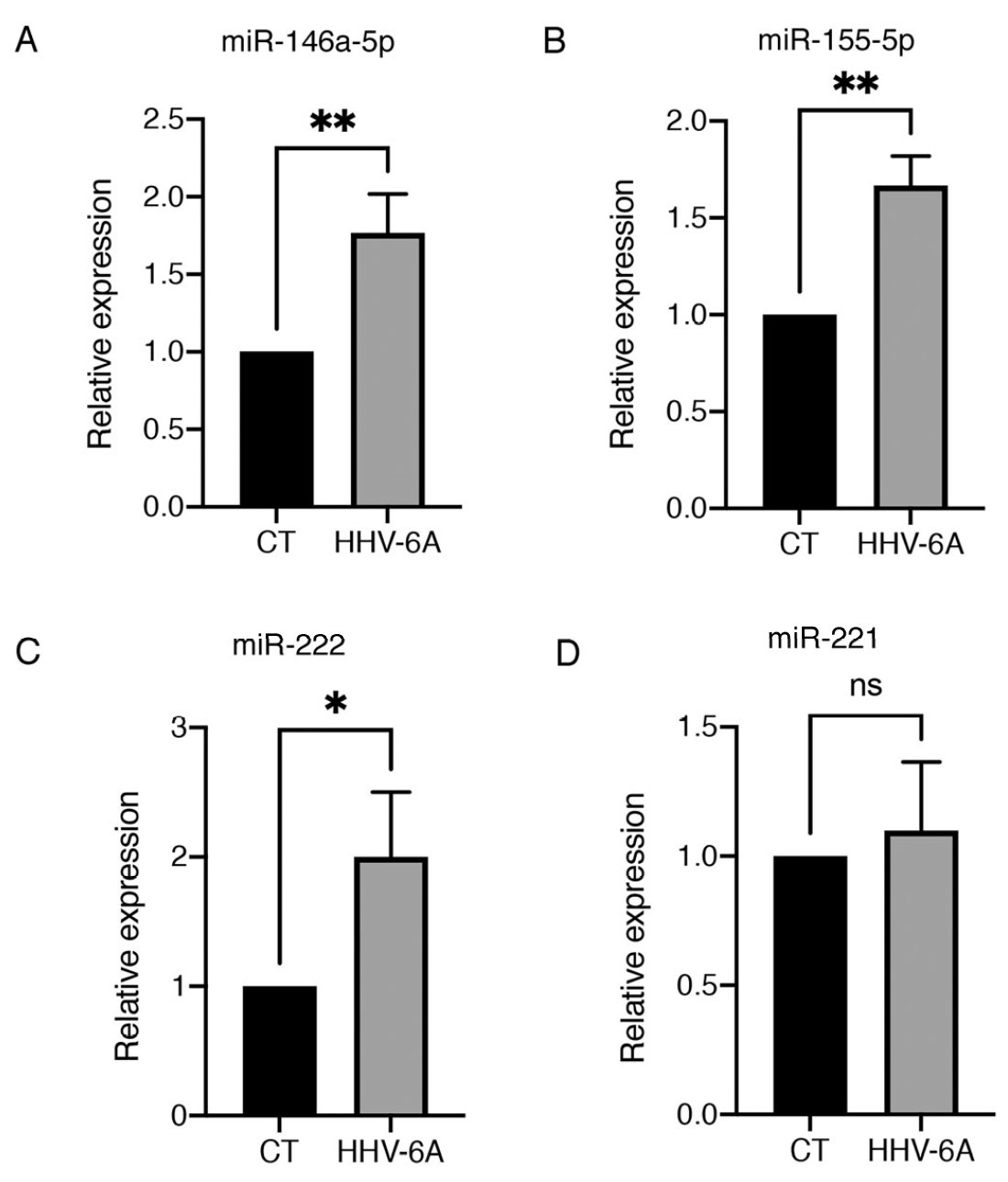
Figure 2: Levels of four miRNAs following mock infection of cells (CT) or following infection with HHV-6A. Relative expression for each microRNA (miRNA) was normalized using U6 small nuclear RNA (snRNA) housekeeping gene. * indicates p < 05, ** indicates p < 0.01.
This interesting study provides support for the possibility that HHV-6A may be a causal agent not only of Hashimoto’s thyroiditis but also of thyroid cancer, which can occasionally evolve from Hashimoto’s thyroiditis since numerous pro-oncogenic changes follow infection of thyroid cancer cells.
Read the full article: Mardente 2024