Persistent HHV-6 infection in the liver is hard to detect from plasma samples and liver biopsies may miss spotty infections. Bile fluid may be the best method for detecting herpesviruses that impact the liver.
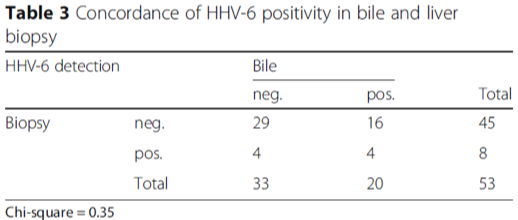 A group from the University Hospital in Heidelberg, Germany performed quantitative polymerase chain reaction for herpesvirus 1–8 DNA in bile, blood, and liver tissue of 73 patients after first liver transplantation (LT). They determined that CMV, EBV, HHV-6, and HHV-7 were all more prevalent in biliary fluid than in liver biopsy tissue or serum. HHV-6 was detected in 34.2% of bile samples while in only 15.1% of biopsy and 2.4% of serum samples. Currently, serum is most often used to test for possible HHV-6 reactivation in transplant patients, but the German study suggests that bile fluid could serve as a more effective marker. The group also found that HHV-6 positivity in bile was significantly associated with allograft complications (including graft rejection and death) but serum positivity was not. This association was found only for HHV-6 and held true even when DNA copies levels were relatively low.
A group from the University Hospital in Heidelberg, Germany performed quantitative polymerase chain reaction for herpesvirus 1–8 DNA in bile, blood, and liver tissue of 73 patients after first liver transplantation (LT). They determined that CMV, EBV, HHV-6, and HHV-7 were all more prevalent in biliary fluid than in liver biopsy tissue or serum. HHV-6 was detected in 34.2% of bile samples while in only 15.1% of biopsy and 2.4% of serum samples. Currently, serum is most often used to test for possible HHV-6 reactivation in transplant patients, but the German study suggests that bile fluid could serve as a more effective marker. The group also found that HHV-6 positivity in bile was significantly associated with allograft complications (including graft rejection and death) but serum positivity was not. This association was found only for HHV-6 and held true even when DNA copies levels were relatively low.
All the patients in this study were treated with prophylactic valganciclovir to prevent CMV infection. While it did prevent CMV complications the prophylactic antiviral did not reduce the incidence of HHV-6 prevalence nor the mean viral count in bile. These results suggest that while prophylactic antiviral treatment for HHV-6 may be effective in lowering the morbidity and mortality of LT patients, the current therapies available are simply not effective enough. Ganciclovir is less effective in vitro against HHV-6 than it is against CMV. The authors conclude that further study is needed to both confirm the efficacy of measuring HHV-6 in bile and to promote the development of HHV-6 specific treatments.
HHV-6 liver disease cannot be ruled out in the absence of a biopsy because in most cases, the infection is localized, without abnormal HHV-6 DNA levels in the blood (Phan 2017, Buyse 2013).
For more information read the full text: Rauber 2019

